Clinical Evidence by Therapeutic Area
The results of research studies utilizing Orthofix products and technologies have been published in a variety of forums, including scholarly journals. Some of these works are highlighted below under their respective therapeutic area.
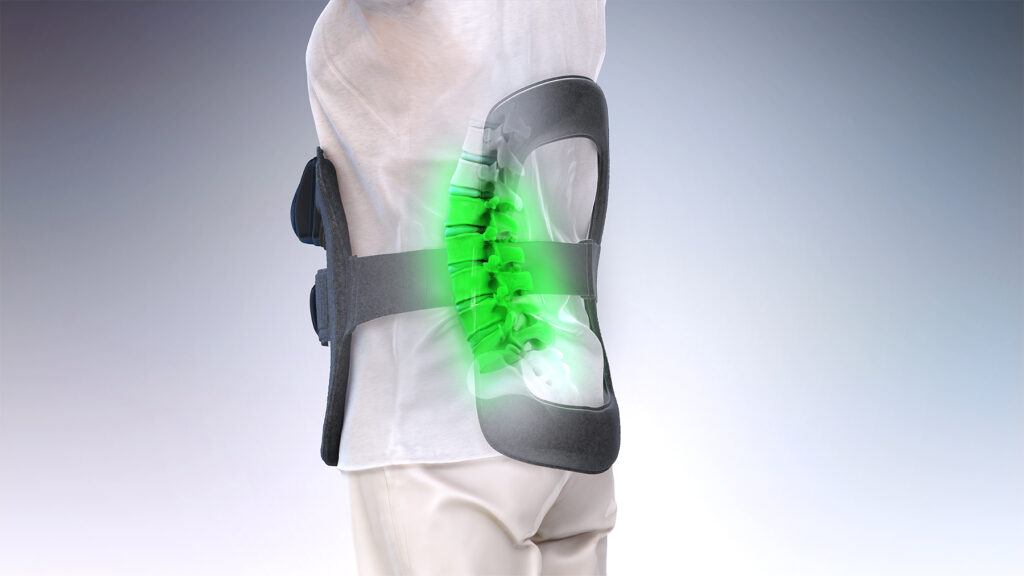
Bone Growth Therapies
With almost 40 years of healing patients, our bone growth therapy devices are the number one prescribed bone growth stimulators in the U.S. These devices provide patients with safe, non-surgical treatment options for promoting spinal fusion and healing nonunion fractures. Using Orthofix’s pulsed electromagnetic field (PEMF) technology, the devices generate a uniform, low-level electrical field that helps activate and augment the body’s natural healing process to enhance bone fusion.
Motion Preservation
Our M6™ artificial discs are the natural choice for helping preserve motion in patients undergoing cervical or lumbar disc replacement. Designed to mimic a patient’s natural vertebral disc, our M6 discs are the only artificial discs that incorporate an artificial nucleus and woven fiber annulus. This design aims to provide the same motion characteristics of a natural disc, including compressive capabilities and a controlled range of motion.

Biologics
Our biologic solutions play an important role in a number of surgical applications, including allografts with viable cells, structural allografts, synthetic bone grafts, and amniotic membranes. By supplying the key elements needed for bone healing coupled with its enhanced handling properties, the Trinity ELITE® and Trinity Evolution™ allografts with viable cells have been implanted in more than 200,000 procedures.
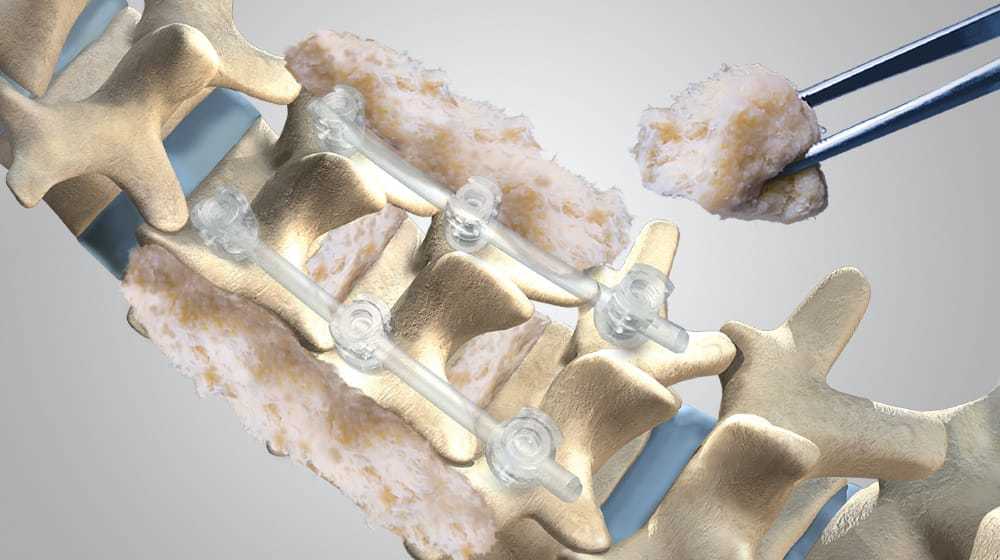
Spine Fixation
We are committed to helping restore quality of life for our patients through advanced interbody and fixation solutions that aid surgeons in repairing spinal alignment, improving disc height, providing nerve decompression and correcting instabilities. Our spine fixation products are used globally in a variety of spine procedures involving deformity correction, minimally invasive approaches, sacroiliac joints, degenerative disc disease, trauma and tumors.

Orthopedics
Orthofix offers innovative and minimally invasive orthopedics solutions to help surgeons improve their patient’s quality of life. Designed to address the lifelong bone and joint health needs of patients of all ages, our orthopedics products help patients achieve a more active and mobile lifestyle when compared to other surgical solutions. Our well-rounded product lines offer comprehensive solutions for pediatrics, limb reconstruction, trauma, and foot & ankle specialties.