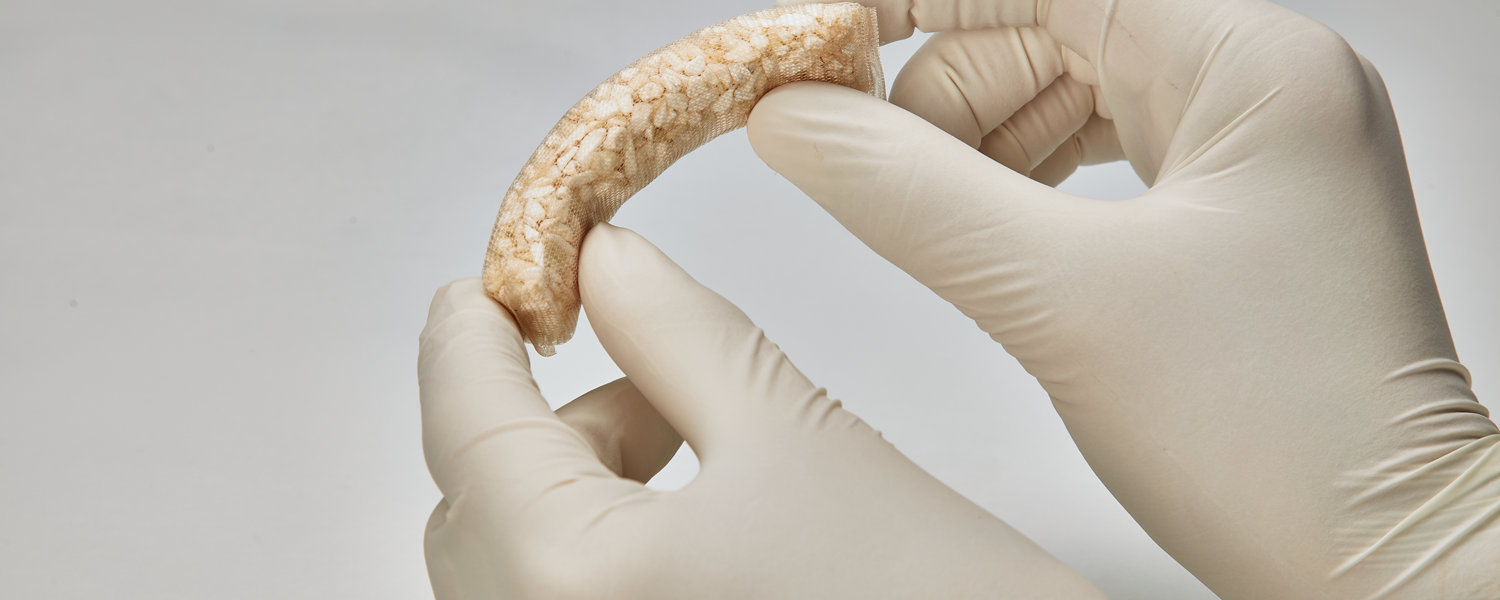
Addressing Graft Migration in Posterolateral Fusion with OsteoBallast™
As spinal fusion procedures continue to rise, driven by a growing incidence of spinal disorders and demand for advanced surgical interventions, the need for solutions that address real procedural challenges has never been greater. One such challenge: bone graft migration in posterolateral lumbar fusion (PLF).
Graft displacement during PLF is a well-documented clinical concern.1 During irrigation or from the compressive forces of the paraspinal muscles during closure, uncontained bone graft can migrate away from the intended fusion site, posing the risk for non-union, fusion at unintended levels, and/or reduced fusion volume.
A Purpose-Built Solution to Graft Migration: OsteoBallast™
OsteoBallast was designed specifically to address graft migration, providing a purpose-built procedural solution for graft containment in PLF.
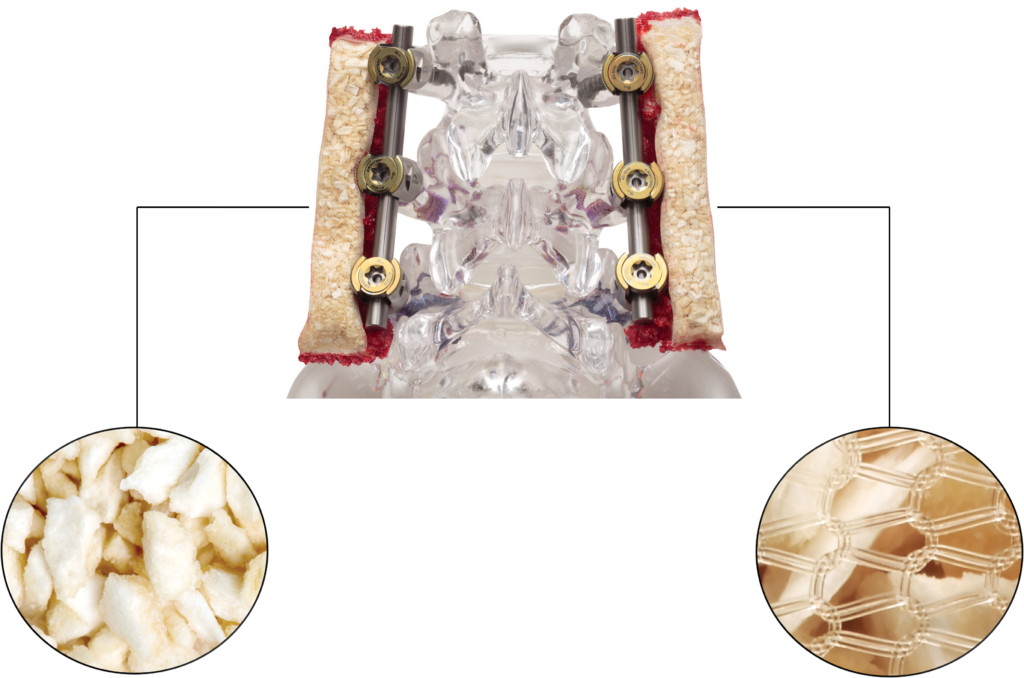
Filled entirely with demineralized bone matrix (DBM) chips, OsteoBallast provides a completely osteoinductive graft. The DBM is contained within a biocompatible mesh pouch, restricting graft migration and maintaining shape while allowing for unimpeded cellular and vascular infiltration essential for bone formation.
Shaped for Maximum Contact with Patient Bone
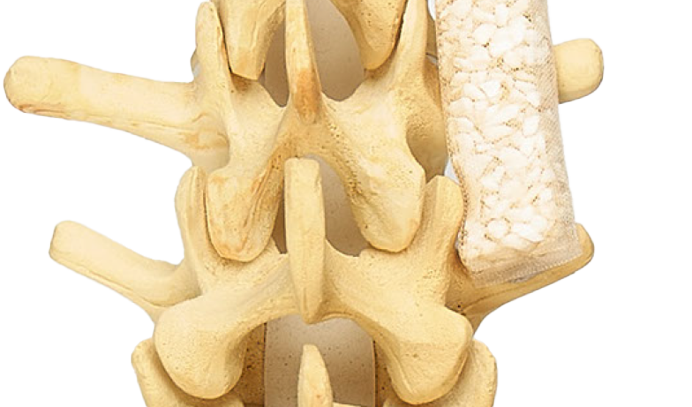
OsteoBallast’s rectangular shape was intentionally designed to maximize surface contact with the transverse processes—the critical contact point for posterolateral fusion. It contours around spinal hardware and anatomy, resists compression, and is simple to deliver and position in the posterolateral gutters.

Improved Surgical Efficiency
With no hydration or preparation required, OsteoBallast supports a more streamlined PLF workflow. Available in five pre-filled sizes, each implant is designed for efficient placement across 1–3 levels, minimizing intraoperative steps and enhancing procedural efficiency.
Flexibility for Surgeon Preferences: OsteoBallast MT
For surgeons who prefer to customize their graft, OsteoBallast MT offers an empty resorbable mesh pouch that can be filled with autograft, trimmed to the desired length, and tailored for specific handling, bringing the benefits of containment to a broader range of surgical preferences.

Evidence of Graft Containment for Enhanced Fusion Outcomes
In two preclinical PLF models, grafts that were contained achieved 100% fusion rates, compared to only 52–60% for uncontained grafts.2-3
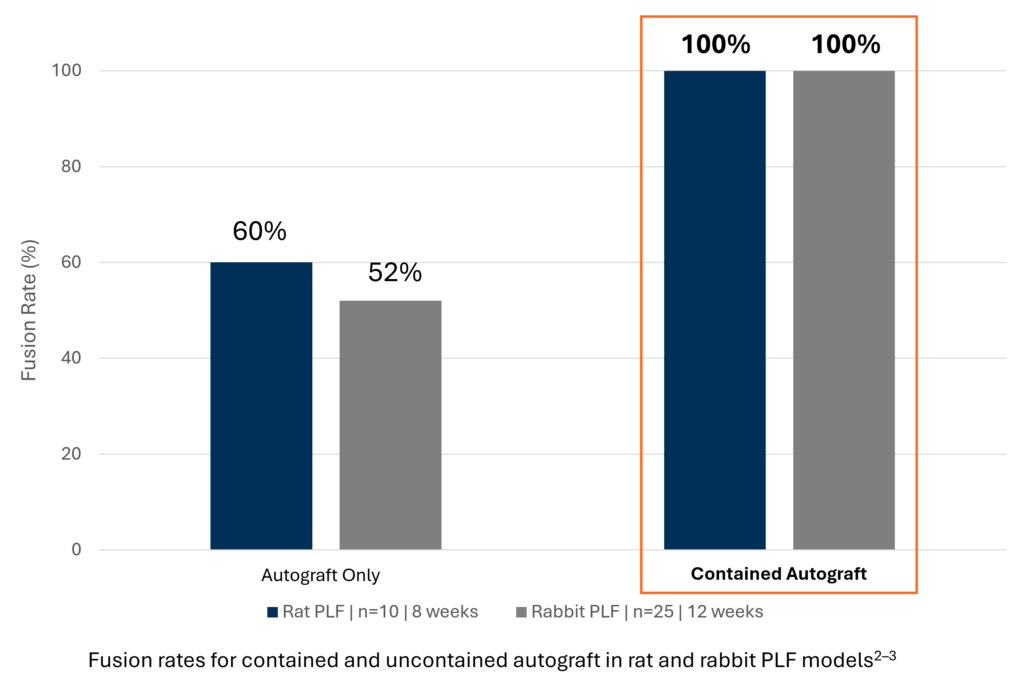
Graft containment has been shown to significantly improve fusion outcomes in PLF across various pre-clinical studies. In two preclinical PLF models, grafts that were contained achieved 100% fusion rates, compared to only 52–60% for uncontained grafts.2-3 Additionally, contained grafts saw a significant increase in fusion mass volume when compared to uncontained groups.3-5
This evidence further underscores the power of graft containment in achieving robust posterolateral fusion outcomes. With a purpose-built design to address graft migration, OsteoBallast keeps graft where it belongs, maximizing PLF fusion potential where it matters most.
To request a demo of OsteoBallast, or to find a sales rep in your area, please contact us here.
To learn more about OsteoBallast and our full portfolio of biologics offerings, visit our webpage.
References
- Bohl M.A., Xu D.S., Daniels L., et al. The Barrow Innovation Center case series: early clinical experience with novel, low-cost techniques for bone graft containment in the posterolateral fusion bed. World Neurosurgery 2018;116:285-95.
- Bawa M., Schimizzi A.L., Leek B., et al. Paraspinal muscle vasculature contributes to posterolateral spinal fusion. Spine 2006; 31:891-896.
- Shin D., Yang B.M., Tae G., et al. Enhanced spinal fusion using a biodegradable porous mesh container in a rat posterolateral spinal fusion model. Spine Journal. 2014; 14:408-15.
- Rao R.D., Bagaria V., Gourab K., et al. Autograft containment in posterolateral spine fusion. T Spine J. 2008; 8:563-69.
- Poynton A.R., Zheng F., Tomin E., et al. Resorbable posterolateral graft containment in a rabbit spinal fusion model. J Neurosurg 2002; 97:460-63.