
Behind the Scenes with R&D: Developing OsteoCove™
“The inspiration behind OsteoCove…Make an engineered material that’s capable of generating bone growth on its own, redefining what a synthetic graft can do.”
OsteoCove is the latest addition of synthetic grafts in the Orthofix biologics portfolio. As an advanced bioactive synthetic, OsteoCove is designed to be more than just a scaffold. Featuring engineered ceramic granules with a microporous surface shown to facilitate bone growth, the next-gen putty and strip formulations are intended to provide reliable handling for application and placement surgeons can trust.
We sat down with Annika Keuther, Sr. Product Development Engineer and research and development lead for OsteoCove, for a behind-the-scenes look at the philosophy, key questions, and data that shaped this advanced bioactive synthetic graft.
Inspiration and Development of an Advanced Bioactive Synthetic
Traditional synthetic grafts have utilized bone-like minerals beta-tricalcium phosphate (β-TCP) and hydroxyapatite (HA) for decades. Combined with collagen, they mimic the composition of bone and provide a biocompatible, osteoconductive scaffold that can be used as a safe and effective bone graft. In recent years, a new class of advanced synthetic grafts have emerged that challenge the status quo.
Q. What was the thought process and the inspiration behind developing OsteoCove?
A. The inspiration behind OsteoCove was to challenge the expectations set by traditional grafts that only act as an osteoconductive scaffold and make an engineered material that’s capable of generating bone growth on its own. Redefining what a synthetic graft can do.
Q. Can you dive into why we chose the components that comprise OsteoCove?
A. Of course. In either case of the putty or strip, the star of OsteoCove is the ceramic granule. We synthesize granules in the lab so they have a biphasic composition—both β-TCP and HA in each granule. Think of how alcohol in water makes a single solution.
We spent several years honing the formulation of this active component aiming for granules that were bioactive, meaning they can spontaneously form apatite in simulated body fluid, like bioactive glass. We then iteratively tested multiple formulations in preclinical models to target our main goal: bone growth.

Q. And how do the putty and strip differ?
A. We decided that these formulations need to be specifically tailored to different applications.
The putty was designed to fill and mold around anatomy and hardware, resist irrigation, and yet not stick to gloves. We chose the reverse phase medium (RPM) carrier we use in Accell™ Evo3 Putty that has received great surgeon feedback. It gels at body temperature, and has a tailored resorption intended to minimize irrigation and graft migration. There’s also type-I bovine collagen in the putty that our surgeon design team agreed enhanced the handling.1
In the case of strip, the goal was to embed the ceramic granules within a collagen scaffold that could span the transverse processes. Again, we didn’t want the ceramic granules migrating once implanted in the body. Cross-linking type-I collagen weaves the granules in and gives the strips the strength needed to resist both compression and tearing.1
Q. How is OsteoCove able to initiate bone growth as opposed to traditional synthetics, even though the components are similar?
A. We targeted the same base components as you mentioned because we know that they’re biocompatible. We then started iterating our manufacturing process to create granules with different shapes and surfaces.
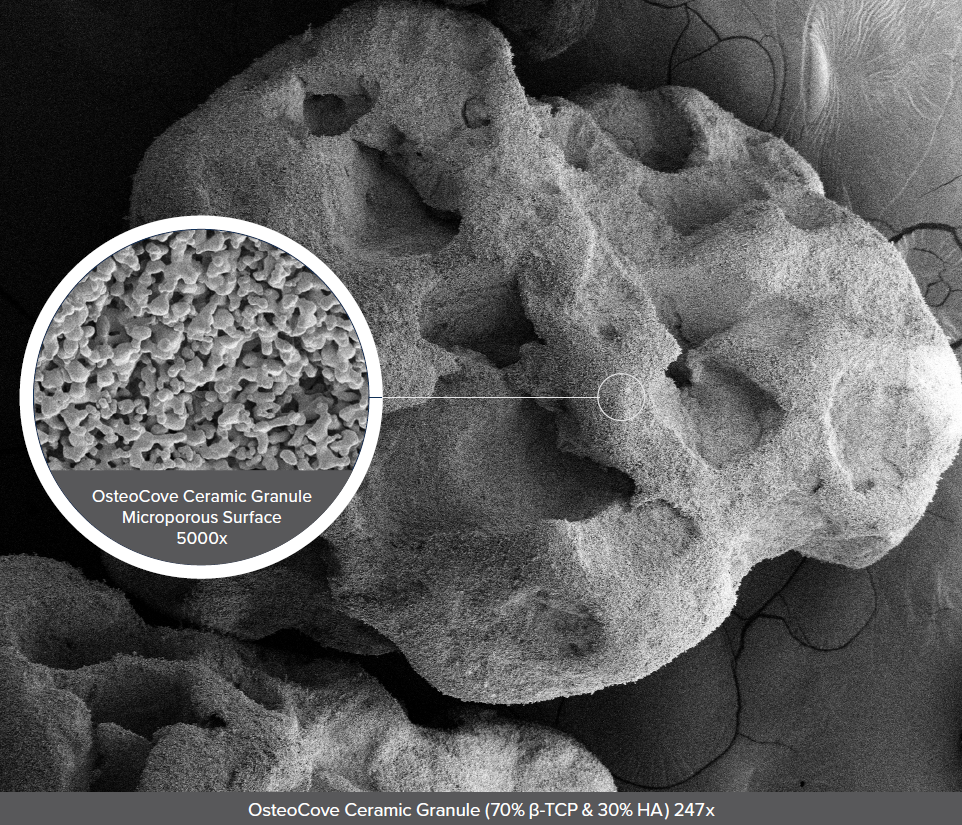
Through that, we generated a specific structure that exists throughout the granules and importantly, on the surface. We can show that these are no longer just scaffolds. The surface is driving a bone forming response, and we’ve used a well-known muscle-pouch model to prove it.
Proving it with Preclinical Data
To validate the graft’s bone-forming performance, OsteoCove was put through one of the more demanding tests for bone grafts: the muscle pouch model—the same conceptual framework first used by Dr. Marshall Urist and widely used to test DBMs for osteoinductivity.
Q. Can you tell us a little bit about what the muscle pouch model is and what makes it so impactful in demonstrating performance?
A. The muscle pouch model is important because it can be used to test if a graft material can generate bone tissue. The reason why material is implanted in the muscle is that there are no bony elements present, and muscles don’t grow bone on their own. If we implant material in there and we see bone formation, we know it was caused by the graft.
“We can show that the surface morphology is what is driving that additional bone formation.”
Typically, either a rat or a sheep model is used. In the case of OsteoCove, we assessed multiple graft materials using a larger sheep model, allowing us to directly compare the performance of different groups in a head-to-head within the same animal.
We compared a traditional synthetic, β-TCP, as a control, to OsteoCove, both with and without the microporous surface. As expected, no bone formation was seen with the traditional synthetic, whereas OsteoCove, with the intact microporous surface, not only generated mineralized bone, but developed bone marrow as well. Demonstrating that it was the surface that led to the formation of well-developed bone tissue.1
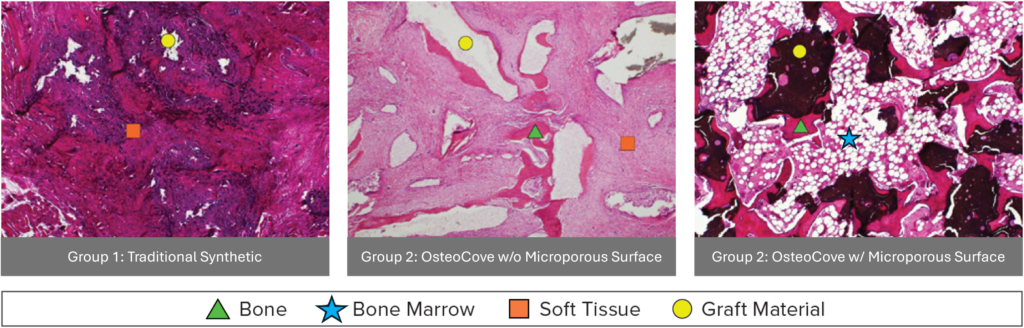
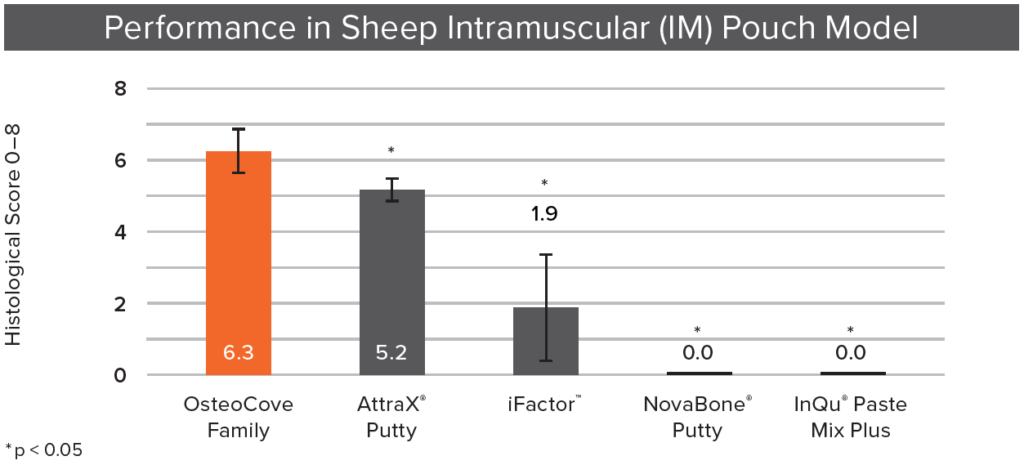
Q. This same model was used to compare commercially available products as well, correct?
A. Yes, using this model we performed head-to-head testing comparing the OsteoCove family with several other competitors.2 Both OsteoCove Putty and OsteoCove Strip showed greater total mineralized bone area and more bone‑forming elements, such as bone marrow, in histological analyses. The OsteoCove family also outperformed a leading advanced synthetic and leading peptide-based graft. Interestingly, one product with a bioactive indication generated no bone at all, demonstrating that a bioactive claim does not guarantee a bone forming response in this model.
Pushing the Boundaries of Engineered Grafts
We thank Annika for taking the time to walk us through the work behind bringing this exceptional graft to market. As she highlighted, OsteoCove is pushing the boundaries of engineered grafts through:
- Microporous surface technology that stimulates a robust bone forming response1
- Demonstrated bone formation in the challenging muscle pouch model2
- Handling characteristics designed to meet real surgical demands1
It’s clear that OsteoCove is backed by a team deeply committed to the process, the data, and the clinicians who rely on this technology.
Click the links below or email biologics@orthofix.com to learn more about OsteoCove and request a sample.
OsteoCove Putty
- Features a reverse phase medium (RPM) carrier designed to resist irrigation
- Includes type-I collagen for a moldable, workable feel
- Ideal for packing, shaping, and filling irregular spaces

OsteoCove Strip
- Often selected for posterolateral fusions or multi‑level cases (with a 30cc long strip format spanning several levels)
- Embeds granules within a cross‑linked collagen scaffold, giving it strength, flexibility, and resistance to compression
- Cross-linked technology with type-I collagen stimulates cell attachment1

References
- Data on File.
- Robertson, S. F., Kuether, A., He, J., Walsh, W. R., & Vizesi, F. (2023). Superior bone formation of OsteoCove™ over competitors in a challenging sheep muscle pouch model (SeaSpine Research White Paper). SeaSpine Orthopedics Corporation.
All other third-party trademarks, service marks, or trade names are the property of their respective owners. Use or display of these third-party trademarks, service marks, or trade names in this document, is not intended to and does not imply any relationship with, endorsement, or sponsorship of these marks by Orthofix.