SpinalStim
Spinal Fusion Therapy
The SpinalStim™ device is FDA approved to be used after spinal fusion surgery or to be used to treat a failed fusion from a previous surgery.1-3 The devices stimulate the natural healing process of bone by sending low-level pulses of electromagnetic energy to the injury or fusion site. The device has an overall clinical success rate of 92% in treating spinal fusion surgery patients.1-3 In addition, the SpinalStim device can be used for treatment of a failed spinal fusion, reducing the need for a revision surgery.1,2
A recently published multicenter study demonstrated that patients treated with SpinalStim as an adjunct to lumbar fusion experienced significantly higher successful fusion rates at 12 months than those who were not.4 SpinalStim also demonstrated statistically significant higher fusion rates across multiple challenging patient subgroups, such as patients with prior failed fusions, older age, multi-level procedures, and an increasing number of risk factors.4
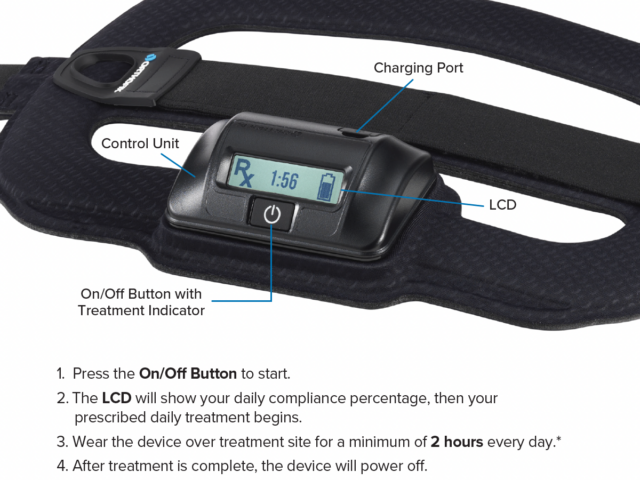
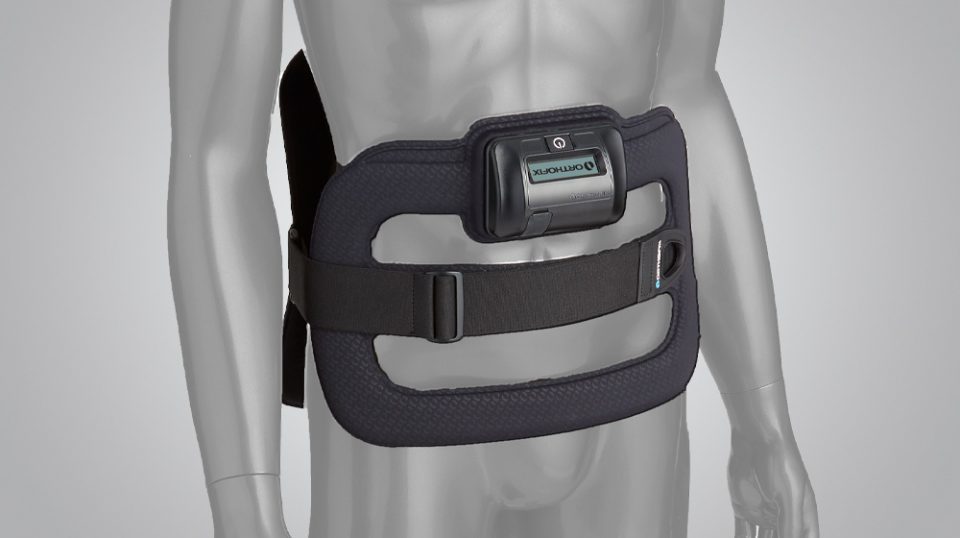
SpinalStim is a single-piece device that is lightweight, flexible and portable, allowing freedom of movement during treatment. The typical prescribed treatment time is two hours per day. An LCD and audible alarm provide important feedback during treatment such as the operational status, treatment time remaining, battery capacity, etc.
Designed for Patient Ease of Use
- Works effectively when worn over clothing or bracing
- Single-piece, cordless design allows for ease of placement and patient mobility
- The STIM onTrack™ app is patient-friendly and provides patients with a treatment calendar, therapy reminders, and additional educational resources.*
Why Do Physicians Prescribe a SpinalStim Device?
- High clinical success rates1-3
- Statistically significant higher fusion rates in patients with risk factors such as multi-level fusion, advanced age, prior failed fusion, smoking, or multiple risk factors.1-4
- PEMF signal covers 360 degrees around the fusion site5
- Coverage up to 5 vertebral levels5
- Supported by the North American Spine Society’s coverage recommendations5
- The STIM onTrack app enables physicians to remotely view patient adherence to prescribed treatments when paired with the STIM MD Platform
For complete prescribing information, please refer to the instruction manual below.
*The STIM onTrack app is available as an accessory of US model devices only
Patient Education Websites
Have Experience With the SpinalStim Device?
Real-world patient experiences can help others better understand what to expect during bone growth therapy.
If you or someone you care for has used the SpinalStim Bone Growth Therapy device, we invite you to share your experience to support patient education and awareness.
Submissions are reviewed, and a signed release is required before any story is shared publicly.
Support Material
Footnotes
*The results of preclinical studies may not be indicative of human clinical trials.
- PMA P850007/S6. February 1990.
- Simmons JW, Mooney V, Thacker I. Pseudarthrosis after lumbar spine fusion: non-operative salvage with pulsed electromagnetic fields. American Journal of Orthopedics, 2004 Jan;33(1):27-30.
- Zborowski M, Androjna C, Waldorff El, Midura RJ 2015 Comparison or therapeutic magnetic stimulation with electric stimulation of spinal column vertebrae. IEEE Transactions on Magnetics 51(12): #5001009, doi: 10.1109/TMAG.2015.2458297
- Hassanzadeh H, Weinstein M, Lansford T, et al. A retrospective, controlled trial evaluation of pulsed electromagnetic field stimulation for fusion success following lumbar spinal surgery. Journal of Spine Surgery, 2026 Jan 31;12(1):2.
- spine.org/PolicyPractice/CoverageRecommendations/AboutCoverageRecommendations.aspx
*Money back guarantee does not apply to wholesale orders.